Using Cold-Stage MDRS to Study Temperature-Driven Particle Changes in Nasal Spray APIs
Related webinars:
For a deeper look at MDRS best practice and how to get the most from the Morphologi 4-ID, watch our webinar on MDRS of complex generics.
Alongside MDRS, our analytical chemistry expertise is reflected in our impurities webinar series covering:
Cold-Stage MDRS Investigation of Beclomethasone Dipropionate:
In Situ Monitoring Morphological and Chemical Stability of Nasal Spray API Across Different Temperatures

Authors:

Dr Jabbar Gardy
Dr Jabbar Gardy is a Technical Leader in Physical Characterisation with a PhD in Chemical Engineering.

Caroline Aiken
Caroline Aiken has been successfully growing and leading the Physical Characterisation team at Bradford for the past 6 years.

Caroline German PhD FRSC
Caroline German is Operations Director at Cormica Bradford with over 25 years’ experience in analytical chemistry, physical characterisation, and GMP.
Share with your colleagues:
What and why we are doing: INTRODUCTION
- Nasal drug delivery is increasingly important due to rapid systemic absorption and avoidance of first-pass metabolism.
- Beconase® nasal spray, as a case study of OINDPs, contains beclomethasone dipropionate (BDP) active pharmaceutical ingredient (API) in an aqueous suspension delivered via a metered-dose actuator.
- Suspension stability requires tight control of particle characteristics during manufacturing, storage, and use.
- Wide global ambient temperature fluctuations during transport/storage has the potential to induce particle size / shape changes in the API of such formulations, potentially altering crystal habit or morphology, and impacting overall product performance if temperature is not adequately controlled.
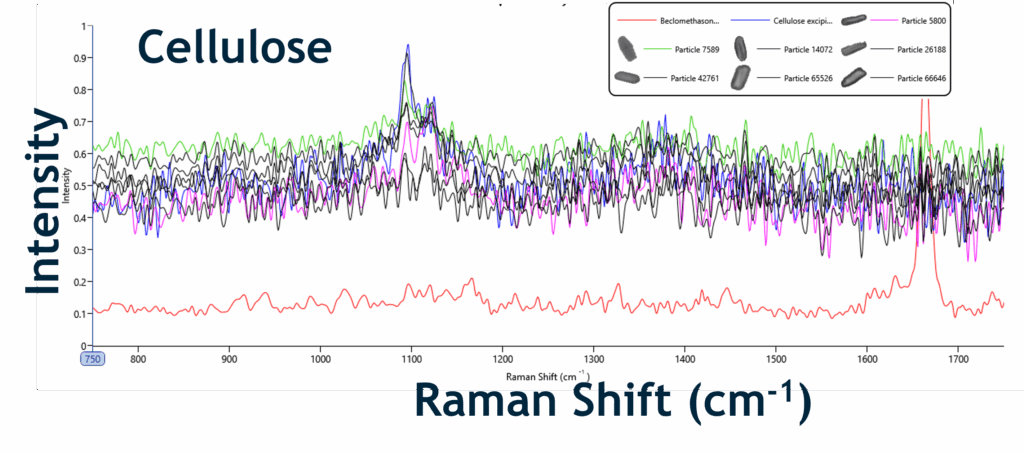
- Morphologically-directed Raman spectroscopy (MDRS) combines automated image analysis + Raman ID = distinguishes API vs excipients.
- Cold-stage MDRS extends this by allowing for the mimicking real-world storage and use conditions during analysis.
Objectives:
- Develop and test a first bespoke cold-stage integrated with MDRS.
- Examine temperature-driven morphological changes of BDP API.
- Assess excipient stability under identical conditions.
How we are doing it: METHODS
- Commercial Beconase® (BDP API 50 µg/spray, Boots Pharmacy, UK) was used as a model nasal spray for analysis.
- Novel custom built cold-stage was coupled with MDRS and used for the inline analysis of particle size, shape and chemical identification of the BDP API and excipients within the Beconase® formulation at different temperatures.
- Sample preparation method was developed and instrumental configuration parameters optimised for this application.
- Investigate the impact of real-world storage and transport conditions on the API and excipients particles within the formulations.


What we have found: RESULTS
Manual Microscopic Images of Captured Particles at Various Temperatures:



Automated Microscopic Images of Captured Particles at Various Temperatures:






Particle Size Distributions of BDP API and Cellulose Excipient Particles:
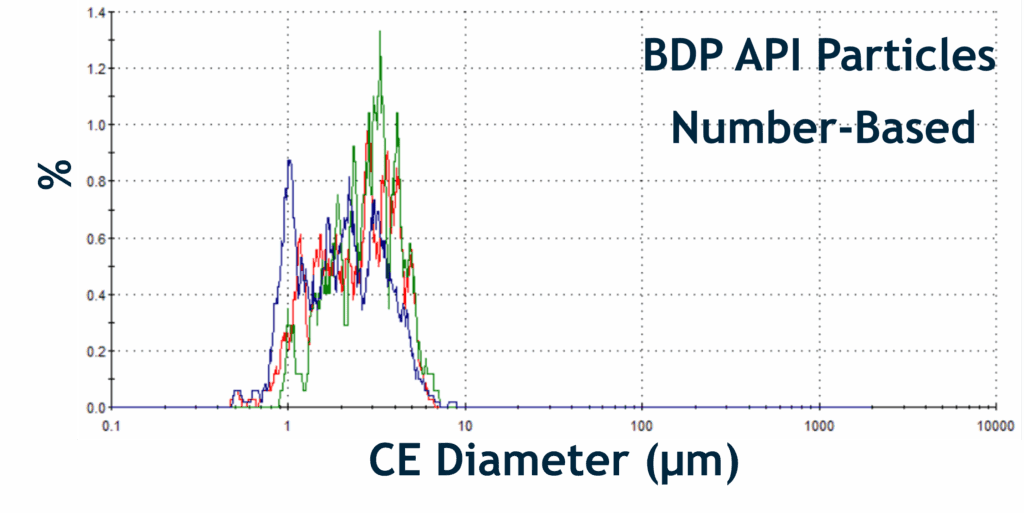

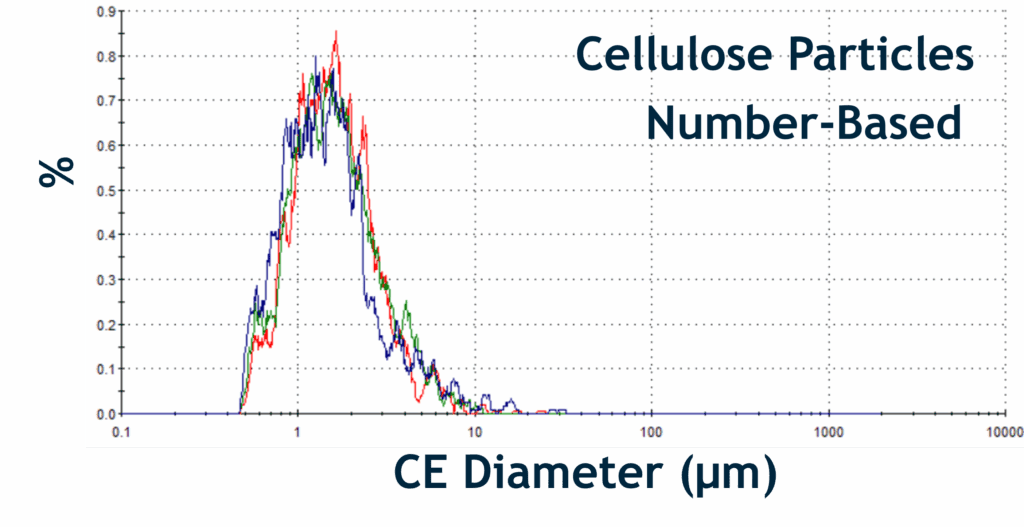

Overlaid Size Distributions of Different Particle Classes within the Beconase at Different Temperatures. Colour-coded Lines: Red Line: 25 °C; Green Line: 37 °C; and Blue Line: 50 °C
Chemical Identifications of Different Particle Classes within the Beconase Nasal Spray:

What it means: CONCLUSIONS
- A novel custom-designed Cold-Stage coupled with MDRS and used for the first time to monitor in situ of temperature-dependent morphological and chemical stability of BDP API and excipient in Beconase® nasal spray formulation.
- It was found that BDP API particles were temperature sensitive, with pronounced morphological changes at elevated temperatures (37 °C), whilst cellulose excipients showed greater structural resilience under the same conditions.
- Observed marked volume expansion of cellulose particles at higher temperatures (50 °C), further supporting their differential response compared with BDP API particles.
- Next stage will focus on low-temperature and inverted storage orientation conditions.
Other Cormica News, Events & Resources
Cormica's Webinars
Recent News
Technical Resources
Laboratories near you, supporting global product success
With accredited laboratories in the UK, EU and US, we support pharmaceutical, medical device and combination product manufacturers worldwide. Local support combined with global quality, delivered by teams who understand your regulatory requirements.


Our Clients’ Experiences




