Physical & Functional Testing for Medical Devices
Medical devices and combination products must demonstrate reliable performance at every stage of development, from early design decisions through to validation, change control, and post-market support. Physical and functional testing plays a critical role in building that evidence, helping teams understand how devices behave under real-world conditions and where risks may emerge.
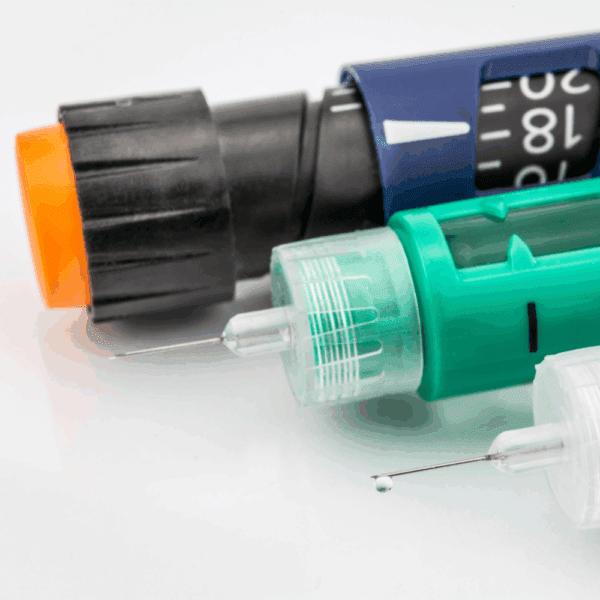
Testing programmes are designed to assess functional performance, mechanical integrity, and usability-related forces in the context of the intended device and its use, aligned with applicable international standards such as ISO 11608 for needle-based injection systems, ISO 11040 for pre-filled syringes, ISO 80369 for small-bore connectors, and ISO 11607 for packaging validation where relevant. This approach supports confident design decisions, robust verification and validation, and clear, regulator-ready data that stands up to scrutiny across global markets.

Types of Medical Devices & Combination Products we Assess Include:
- Pre-Filled Syringes
- Auto-Injectors
- Ophthalmology products
- On-Body & Wearables
- Surgical Equipment
- Anaesthesia Devices
- Dental implants
- Orthopaedics
- Robotic Surgery
- Cardiovascular Devices
Device Verification and Validation (DVT)
Effective physical testing is not just about generating results. It is about designing studies that demonstrate, with confidence, that a device performs as intended under defined conditions of use. Device Verification and Validation (DVT) provides a structured framework for translating design requirements and risk controls into meaningful, regulator-ready evidence.

The timeline for physical and mechanical testing of medical devices depends on the specific tests required, such as tensile, compression, or flow rate tests. We offer flexible testing schedules, with standard testing turnaround times ranging from 1-2 weeks.
Mechanical testing and functional testing is crucial for a wide variety of medical devices, including needle injection systems, drug delivery devices, parental containers, and implantable technologies. Cormica specialises in testing to ensure device durability, performance, and compliance with industry standards like ISO 11608, ISO 11040, and USP 788.
For mechanical testing of medical devices, we follow several key ISO standards, including ISO 11608 for needle-based systems, ISO 11040 for prefilled syringes, and ISO 80369 for small-bore connectors. Our adherence to these international standards ensures your device meets global regulatory requirements.
Design verification testing ensures your medical device is built according to design specifications, while validation testing confirms it meets user needs and regulatory standards. At Cormica, we offer comprehensive physical and mechanical testing to support both design verification and validation processes, helping ensure your device is safe, effective, and compliant.
Laboratories near you, supporting global product success
With accredited laboratories in the UK, EU and US, we support pharmaceutical, medical device and combination product manufacturers worldwide. Local support combined with global quality, delivered by teams who understand your regulatory requirements.

