ISO 18562-3:2024 Carbonyl Testing Requirements and What Manufacturers Need to Know
Evolving requirements for breathing gas pathway testing place new pressure on manufacturers. Cormica provides the carbonyl testing expertise, method development and regulatory guidance needed to transition smoothly to ISO 18562-3:2024. We help you generate clear, compliant data that supports faster submissions and reduces costly project delays.
Are you up to date with the new changes of ISO 18562-3:2024 Biocompatibility evaluation of breathing gas pathways in healthcare applications Part 3: Tests for emissions of volatile organic substances?
From the 5th of July 2026, the U.S. Food and Drug Administration (FDA) will no longer accept test study submissions based on the previous 2017 version of ISO 18562 (2017), they must comply with the current version (2024).
Compared with the previous edition of ISO 18562-3 (2017) containing normative reference to ISO 16000, part 6 only, the newest edition additionally references parts 3 & 4. With this, the requirement for the specific assessments of formaldehyde and other carbonyl compounds is now compulsory.

Authors:

James Silk
Senior Analytical Chemist at Cormica, specialises in extractables and leachables (E&L) testing for medical devices and combination products. With expertise in GC-MS and LC-MS techniques, James supports regulatory submissions through method development, validation, and interpretation of E&L data for global clients.
Jackie Waller
Jackie Waller is Senior Analytical Chemist for Cormica. With a first-class honours degree in Forensic Chemistry, she carries over ten years’ experience working in analytical science, including pesticide residue analysis and small molecule impurity testing of pharmaceutical products. Her current role is dedicated to designing and carrying out analytical studies. Predominantly this relates to extractables and leachables of medical devices and includes residual compound and impurity testing.
Share with your colleagues:
Why is it important to monitor the emission of carbonyls and how can they be released by any medical device?
Many carbonyl compounds have been recognised to pose significant health risks. Their effects can range from mucous membrane irritation to carcinogenicity. Evidence also exists to suggest that these compounds may lead to long-term effects on the nervous system.¹
The presence and release of carbonyl compounds from medical devices may be associated with residues from sterilization, polymer degradation and adhesive/ solvent evaporation.¹ Alongside their volatile nature, the addition of air flow to the material used for medical breathing devices may promote their release into the gas pathway. Temperature effects may also promote their release, for example in the case of a temperature-controlled incubator.
Continuous monitoring ensures any such by-products are identified and mitigated. Breathing devices deliver air directly into the lungs, bypassing some of the body’s natural filters. Any volatile or semi-volatile carbonyl compounds emitted from device materials can be inhaled immediately, posing a high exposure risk.
Cormica are up to date with the required method of analysis for carbonyl compounds including formaldehyde. We have incorporated this additional, specific methodology and testing as part of our existing broad scope VOS testing method.
Analytical Assessment and Methodology
Using state of the art equipment and technology, Cormica can effectively simulate the clinical use of medical devices designed to deliver gas to a patient. In doing so, the gas delivery system can be tested in its entirety for the release of volatile organic substances.
A gas delivery system is often comprised of multiple components. This may include ventilators, breathing tubes, in-line filters, masks and more. All parts possess their own potential for volatile organic substance release and contribution to the airway of the patient.
Based on the volatility and size of formaldehyde and other carbonyl compounds, traditional VOS capture techniques may prove unsuitable. The potential for losses throughout the capture process can significantly decrease the efficiency and effectiveness of the analytical process.
Additionally, gas chromatography analysis may prove difficult with limited column retention. With this in mind, derivatisation of this specific class of compounds is considered an effective solution and can be carried out at the point of release.
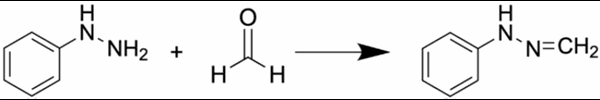
By utilising the favourable reaction of 2,4-dinitrophenylhydrazine (DNPH) with aldehydes and ketones, VOS’s with these specific functional groups can be captured and converted to assist in effective analysis. By simulating the device set up and subjecting it to exaggerated clinically relevant conditions, VOS release is promoted.
By installing DNPH containing cartridges within the gas pathway of the simulated test set up, beyond the medical device, any released carbonyl containing compounds are captured and converted to their DNPH derivatised form.
Using an Agilent 1290 infinity 2, Cormica have developed a LC-UV method capable of fully resolving and detecting 13 DNPH derivatised compounds.
This method allows for the confident assessment of carbonyl containing compounds between C¹-C¹² and with its enhanced sensitivity, the Agilent 1290 infinity 2 allows detection down to concentrations as low as 10 parts per billion (ppb). This supports additional confidence in our ability to detect compounds released from a range of materials often used in medical device production and enhances contribution to patient safety.

Figure 3 displays the range of concentrations of all 13 DNPH derivatised carbonyl compounds from 50ppb to 20ppm, with a Pearson correlation coefficient value of >0.99 for all compounds. Preliminary validation data shows relative standard deviation (% RSD) value <1% was achieved for all compounds. Limit of detection (LOD) was established as 10ppb with a defined signal to noise ratio ≥3 and limit of quantitation for each compound established as three times the LOD. Based on this information, the linear range of 50ppb to 20ppm was selected.
Supporting ISO 18562-3:2024 compliance and patient safety
By integrating specific carbonyl testing into its VOS methodology, Cormica helps manufacturers:
- Align with the updated requirements of ISO 18562-3:2024
- Generate data suitable for FDA submissions beyond July 2026
- Understand carbonyl contributions from each device component and the complete system
- Strengthen risk management for breathing gas pathways and related devices
Talk to us about ISO 18562-3:2024 testing
If you are developing or updating devices with breathing gas pathways and need to understand how ISO 18562-3:2024 impacts your testing strategy, Cormica can help.
¹ISO 18562-3:2024 – Biocompatibility evaluation of breathing gas pathways in healthcare applications. Tests for emissions of volatile organic substances
²Lipskerov, Fedor & Sheshukova, Ekaterina & Komarova, Tatiana. (2022). Approaches to Formaldehyde Measurement: From Liquid Biological Samples to Cells and Organisms. International Journal of Molecular Sciences. 23. 6642. 10.3390/ijms23126642.
Other Cormica News, Events & Resources
Cormica's Webinars
Recent News
Technical Resources
Laboratories near you, supporting global product success
With accredited laboratories in the UK, EU and US, we support pharmaceutical, medical device and combination product manufacturers worldwide. Local support combined with global quality, delivered by teams who understand your regulatory requirements.


Our Clients’ Experiences




