Poster Guide - Plunger Movement in Prefilled Syringes: An Altitude Simulation Study
Understanding How Air Transport Conditions Affect Syringe Integrity
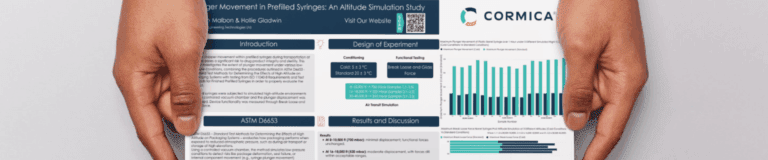
Plunger movement during transportation poses a critical challenge for prefilled syringes, where even slight shifts can compromise sterility, dose accuracy, and product performance. This study explores how simulated high-altitude and temperature conditions influence plunger displacement and syringe functionality.
By combining ASTM D6653 altitude simulation methods with ISO 11040-8 performance testing, the team evaluated syringe behaviour under realistic air transit pressures, ranging from 8,000 ft to 40,000 ft, and across cold chain and standard temperature conditions.
The findings reveal how altitude and temperature interplay to affect glide force, break loose force, and plunger displacement, providing essential insights for design verification, stability, and transport validation of prefilled systems.
Key Topics Covered
- High-altitude simulation testing for medical packaging and syringes
- Impact of cold chain conditions on elastomer performance
- ASTM D6653 and ISO 11040-8 combined testing approach
- Strategies to minimise risk to sterility and dose delivery
See our other valuable poster guides to streamline pre-filled syringe testing processes:
Authors:

Steven Malbon
Steven is Combination & Physical Devices Manager at MET, A Cormica Lab.

Hollie Gladwin
Hollie is a Technical Scientist in the Physical and Combinations department at Cormica MET.
Contact us:
We will email you the link to the PDF file.
Related resources
Cormica's Webinars
Recent News
Technical Resources
Laboratories near you, supporting global product success
With accredited laboratories in the UK, EU and US, we support pharmaceutical, medical device and combination product manufacturers worldwide. Local support combined with global quality, delivered by teams who understand your regulatory requirements.


Our Clients’ Experiences