Sterility Testing
Home » Services » Microbiology Testing » Sterility Testing
Sterility testing supporting batch release, validation and global regulatory submissions.
- Compendial sterility testing to USP <71>, ISO 11737-2, Ph Eur and JP requirements
- Rapid sterility testing options to support faster release timelines
- Experience with complex, high-value and combination products
- Laboratories in the UK, EU and US supporting global programmes
- Investigation support for sterility test failures and OOS results
Sterility testing plays a critical role in product release and regulatory approval, so confidence in your testing partner matters. Cormica’s decades of microbiology experience, specialist scientists and a global laboratory network help support complex programmes, investigations and multi-region submissions for pharmaceutical, medical device and combination product manufacturers worldwide.
GMP | GLP | ISO 17025
Pharmaceuticals
Injectables, parenterals and sterile drug products requiring batch release testing.
Medical Devices
Implants, surgical instruments, wound care products and sterile single-use devices.
Combination Products
Pre-filled syringes, auto-injectors and drug-device combination products.
Biologics & Advanced Therapies
Vaccines, cell and gene therapies and high-value sterile products.
Ophthalmic & Respiratory
Sterile eye products, inhalation and nasal drug delivery systems.
Catheters & Fluid Path Products
IV sets, tubing, dialysis products and fluid transfer systems.
Sterility Testing Methods
- Sterility testing (including Millipore Steritest™)
- Rapid sterility testing
- Celsis Bioluminescence
- Celsis Advance & Celsis Adapt
- Scan RDI Chemiluminescence
- Celsis Bioluminescence
- Particulate testing
- Container closure integrity testing (CCIT)
- Media fill incubation and analysis
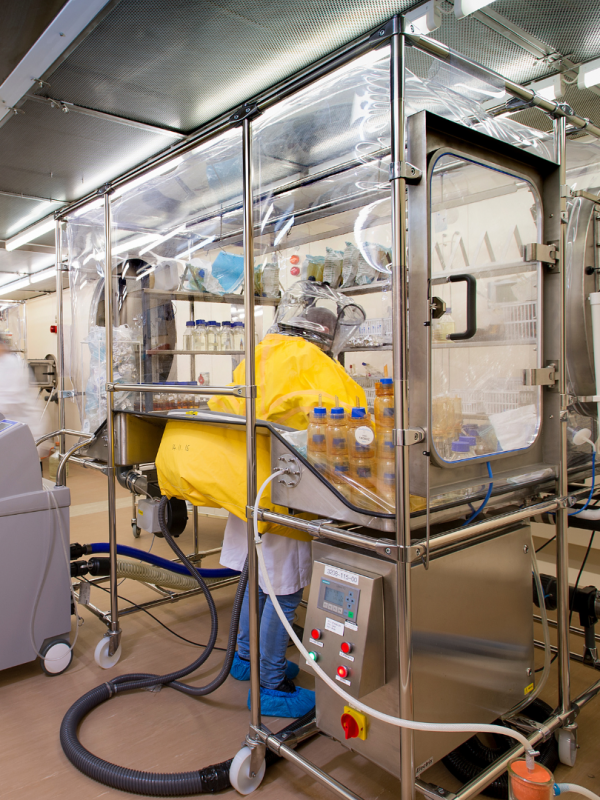
Our facilities operate an (ISO class 7 compliant cleanroom, which offer a number of large capacity half-suit isolators, enabling us to conduct testing on a multitude of different medical devices, combination products, pharmaceuticals and much more.


This method is for samples which are unable to be filtered through a filter membrane with a pore size of 0.45 µm. This method involves transferring the product directly into two types of culture media: Tryptic Soy Broth (TSB) for detecting fungi and aerobic bacteria, and Fluid Thioglycollate Medium (FTM) for anaerobic bacteria. The samples are incubated at appropriate temperatures for 14 days. Any turbidity or change in the media may microbial growth, which signifies contamination.
Ideal for parenterals, ophthalmic solutions, and other aqueous products that can be filtered. WML provide two types of membrane filtration. Traditional open funnel and steritestTM. The product is passed through a sterile membrane filter with a pore size of 0.45 µm, which traps any contaminating microorganisms that may be present within the product. The filter is contained within the appropriate growth medium ( TSB and FTM) and is incubated temperatures for 14 days to detect any microbial contamination. This method is particularly useful for products with antimicrobial properties as appropriate washing steps are performed to eliminate any antimicrobial properties present.
To ensure the chosen method works effectively with the specific product, WML perform method suitability testing for both Direct Inoculation and membrane filtration methods. The purpose of this test is to determine if aerobic and anaerobic organisms will grow in the test after antimicrobial agents have been neutralised. It is recommended the Method Suitability Test is carried out for all products tested.
Laboratories near you, supporting global product success
With accredited laboratories in the UK, EU and US, we support pharmaceutical, medical device and combination product manufacturers worldwide. Local support combined with global quality, delivered by teams who understand your regulatory requirements…
Sterility Testing Frequently Asked Questions
Traditional sterility testing typically takes 14 days to complete. This includes the time required for incubation and observation for any signs of microbial growth. We offer expedited testing services to meet urgent needs.
Sterility testing is required for a wide range of products, including pharmaceuticals (such as injectables and ophthalmic solutions), medical devices (such as surgical instruments and implants), and biologics (such as vaccines and cell therapies).
Cormica follows stringent protocols and use validated methods to ensure the accuracy of sterility testing. Our tests are performed in half suit isolators, which provide a controlled and sterile environment to minimize contamination. Additionally, we implement rigorous quality control measures and continuous monitoring to ensure reliable results.
If a product fails sterility testing, it triggers a thorough out of specification investigation to identify the source of contamination. The result will be a failure if no root cause can be identified.
Sterility testing is carried out on a select few containers from an entire batch. The sterility test itself confirms the containers tested are free from any contaminating microorganisms. The Ph.Eur 2.6.1 states that ‘ a satisfactory result only indicates that no contaminating micro-organisms were found in the conditions of the test.
Non-compliance can lead to:
- Patient safety risks: Increased chances of infections, adverse reactions, or fatalities from contaminated products.
- Legal repercussions: Fines, legal liabilities, and potential litigation.
- Product recalls: Financial losses, brand damage, and disrupted supply chains.
- Market delays: Delayed product launches and missed revenue opportunities.
Compliance ensures patient safety, regulatory approval, and business continuity.
This international standard specifies the requirements for sterility testing of medical devices. It provides guidelines for the preparation, testing, and interpretation of sterility test results, ensuring that medical devices are free from contaminating microorganisms.