On-Body Injector & Wearable Device Testing
Home » Services » Medical Device Testing » On-Body Injector & Wearable Device Testing
On-body delivery systems, patch pumps, and connected wearable injectors must perform reliably over extended wear periods while delivering accurate doses in real-world conditions. These complex drug-device combination products require robust testing to demonstrate performance, usability, and long-term reliability throughout development and commercial production.
Testing programmes for on-body and wearable injectors evaluate adhesion, wear performance, dose delivery, and device reliability. Alignment with relevant parts of ISO 11608 and IEC usability guidance helps generate robust, regulator-ready data to support global submissions.
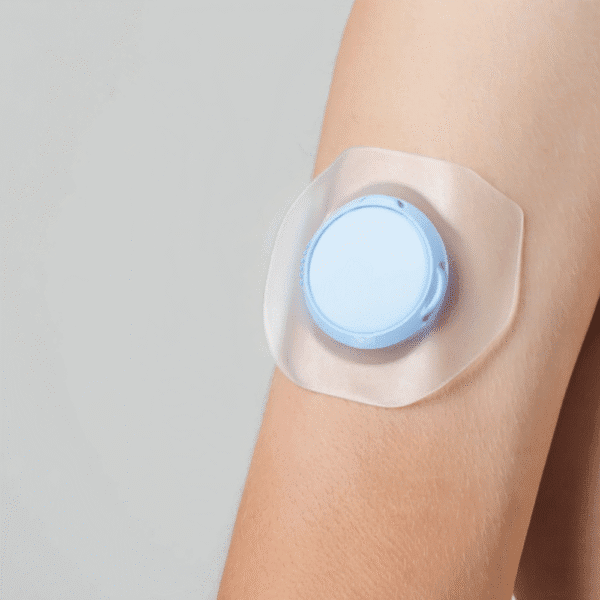

Why testing is critical for on-body and wearable injectors
Wearable injectors are designed for extended contact with the body and often operate outside clinical environments. This introduces additional challenges related to adhesion, usability, reliability, and long-term performance.
Key challenges include:
- Maintaining reliable adhesion during extended wear
- Delivering accurate and consistent doses over time
- Demonstrating device reliability in real-world conditions
- Ensuring usability and safe operation by patients
- Managing environmental and lifecycle impacts on performance
- Supporting global regulatory expectations for combination products
Comprehensive testing helps identify risks early and generate the evidence required for regulatory approval.
On-body injector testing services across the device lifecycle
Analytical Chemistry
Material characterisation, extractables and leachables studies, and impurity analysis support product safety.
Microbiology & Sterility
Bioburden, sterility, and endotoxin testing support microbiological safety for combination products.
Physical & Packaging Testing
Wear testing, adhesion assessment, dose delivery evaluation, usability testing, and reliability studies support device performance.
Biocompatibility & Toxicology
ISO 10993 biological evaluation and toxicological risk assessment support safe patient contact.
Consultancy & Scientific Support
Testing strategies and submission support help navigate global regulatory pathways.
Supporting every stage of the On-body injector devices lifecycle
-
Research & Early Development
Material selection and feasibility studies help assess adhesion, wear performance, and device feasibility.
-
Design Verification
Testing confirms device performance, adhesion, dose delivery, and reliability meet design inputs.
-
Design Validation
Evidence is generated to demonstrate safe and effective real-world use.
-
Regulatory Submission & Approval
Testing data supports global regulatory submissions and compliance with recognised standards.
-
Commercial Production & Post-Market Support
Ongoing testing supports change management, lifecycle compliance, and product improvements.
Wearable injector standards and regulatory expectations
Testing programmes for on-body injectors and wearable devices are typically aligned with:
- Relevant parts of ISO 11608 for needle-based injection systems
- IEC usability guidance for medical devices
Alignment with recognised standards helps ensure robust, regulator-ready data for global submissions.
Partner with Cormica
Integrated testing programmes bring together expertise across microbiology, analytical chemistry, physical testing, and biocompatibility to support combination product development from concept through to commercialisation. Decades of experience across hundreds of device programmes worldwide help teams generate reliable data and move forward with confidence.
Laboratories near you, supporting global product success
With accredited laboratories in the UK, EU and US, we support pharmaceutical, medical device and combination product manufacturers worldwide. Local support combined with global quality, delivered by teams who understand your regulatory requirements.


Our Clients’ Experiences




